Date: December 2018 E. Dumont
We first explore the level of theory suited to reproduce the electron affinity and optical properties of the electron attachment on disulfide. During a second practical, we will investigate the same "reaction" within protein relying on a QM/MM approach. 1. Specific requirements for a correct basis set ?
In passing, feel free to identify the various steps of the Gaussian calculation with the reference to the various links, whose specification is given on Gaussian website online, (e.g. /softs/gaussian/g03a02/g09/l1.exe, which you can search Link in the output file). - (Enter /softs/gaussian/g03a02/g09/l101.exe) Reads title and molecule specification - (Enter /softs/gaussian/g03a02/g09/l202.exe) Reorients coordinates, calculates symmetry, and checks variables - (Enter /softs/gaussian/g03a02/g09/l301.exe) Generates basis set information - (Enter /softs/gaussian/g03a02/g09/l302.exe) Calculates overlap, kinetic, and potential integrals - (Enter /softs/gaussian/g03a02/g09/l303.exe) Calculates multipole integrals - (Enter /softs/gaussian/g03a02/g09/l401.exe) Forms the initial MO guess - (Enter /softs/gaussian/g03a02/g09/l502.exe) Iteratively solves the SCF equations - (Enter /softs/gaussian/g03a02/g09/l601.exe) Population and related analyses - (Enter /softs/gaussian/g03a02/g09/l9999.exe) Finalizes calculation and output As an aparte, one can also notes that in some QM software, basis sets are not localized but described as plane waves, which avoids the awful acronyms and the lack of systematic behavior. On the other hand, here one could choose to treat the sulfurs with diffuse functions to save a bit the computation time. 2. Three-electron two-center bonding: a test case for DFT. DFT comes with a rather accurate treatment of electron correlation which can trigger some ameliorations to our QM description of 2S-3e. On the other hand, density functionals are fitted on even-number systems and radicals often requires a more careful description. Also radical systems tend to be more multi-reference. For this system, one can compute a T1 diagnostic to ensure the mono-referential description makes sense.
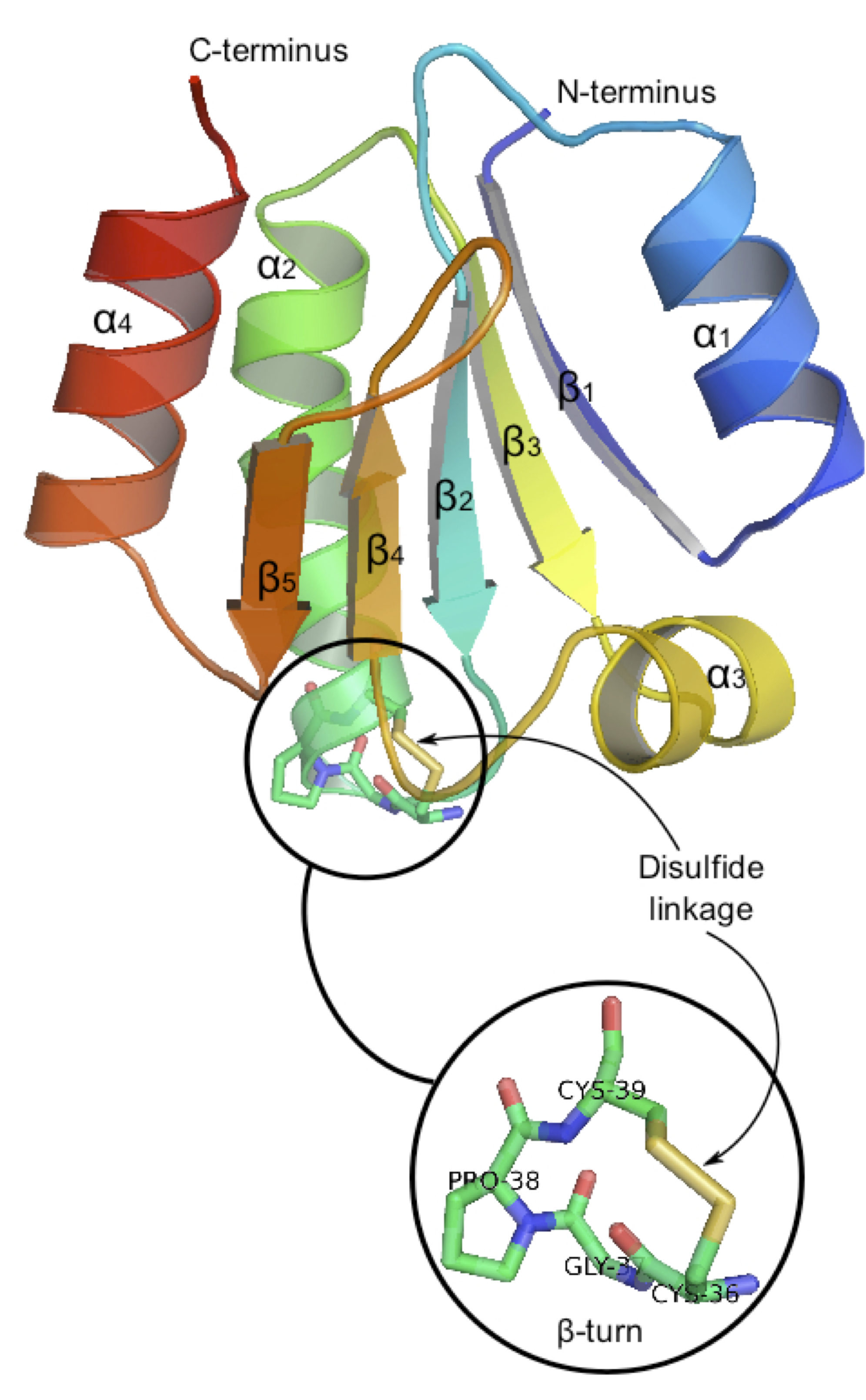
3. Solvation Within proteins, disulfide bridges are often strongly solvent-exposed (to water). We are going to test the several implicit models (before QM/MM...).
Each group can try: scrf=pcm, SCRF=(Dipole,solvent=water,A0=3.71) (the Onsager model), SCRF=smd (Truhlar's model), SCRF=(sas) to compare several approaches. 4. Optical properties The identification of 2S-3e in biological molecules relies on a characteristic UV-Vis signature σ -> σ* at 410nm in water.
Results Results HF+DFT (notebook by the student)
H 0
S 3 1.00
19.2406000 0.0328280
2.8992000 0.2312080
0.6534000 0.8172380
S 1 1.00
0.1776000 1.0000000
P 1 1.00
1.0000000 1.0000000
P 1 1.00
0.7500000 1.0000000
S 1 1.00
0.0441500 1.0000000
****
C 0
S 6 1.00
4232.6100000 0.0020290
634.8820000 0.0155350
146.0970000 0.0754110
42.4974000 0.2571210
14.1892000 0.5965550
1.9666000 0.2425170
S 1 1.00
5.1477000 1.0000000
S 1 1.00
0.4962000 1.0000000
S 1 1.00
0.1533000 1.0000000
P 4 1.00
18.1557000 0.0185340
3.9864000 0.1154420
1.1429000 0.3862060
0.3594000 0.6400890
P 1 1.00
0.1146000 1.0000000
D 1 1.00
0.7500000 1.0000000
S 1 1.00
0.0430200 1.0000000
P 1 1.00
0.0362900 1.0000000
****
S 0
S 5 1.00
35710.0000000 0.0025650
5397.0000000 0.0194050
1250.0000000 0.0955950
359.9000000 0.3457930
119.2000000 0.6357940
S 3 1.00
119.2000000 0.1300960
43.9800000 0.6513010
17.6300000 0.2719550
S 1 1.00
5.4200000 1.0000000
S 1 1.00
2.0740000 1.0000000
S 1 1.00
0.4246000 1.0000000
S 1 1.00
0.1519000 1.0000000
P 4 1.00
212.9000000 0.0140910
49.6000000 0.0966850
15.5200000 0.3238740
5.4760000 0.6917560
P 2 1.00
5.4760000 -0.6267370
2.0440000 1.3770510
P 1 1.00
0.5218000 1.0000000
P 1 1.00
0.1506000 1.0000000
D 1 1.00
0.7000000 1.0000000
S 1 1.00
0.0426700 1.0000000
P 1 1.00
0.0409600 1.0000000
****
Last update : Dec. 2018 |