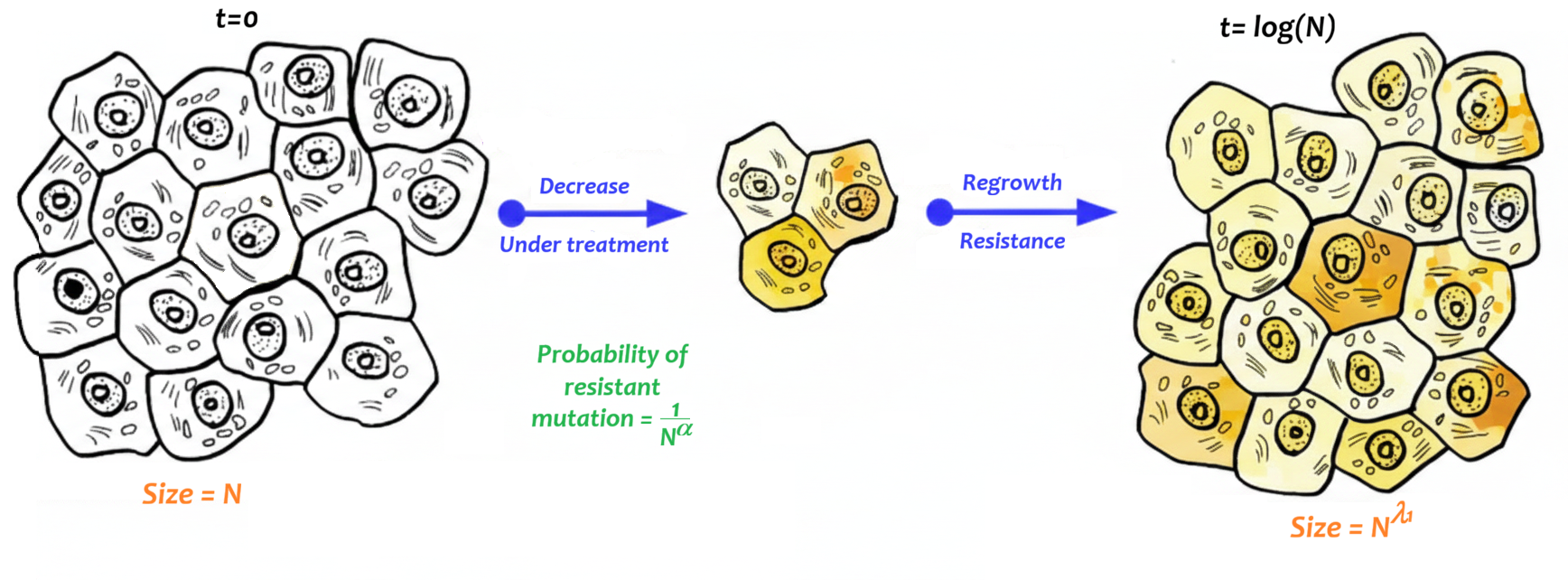
This project aims to model the dynamics of cell populations that
experience "rescue" events, where cells initially sensitive to
treatment mutate, become resistant, and start to grow despite continued
therapy. This phenomenon is commonly observed in cancer, where
treatment resistance emerges over time due to genetic mutations,
leading to a subpopulation that survives and proliferates.
The
objective of this project is thus to investigate the composition and
behavior of a cell population undergoing such event, by developing and
analyzing stochastic models that represent these dynamics, aiming to
gain deeper insights into how resistance develops and spreads.
More presicely, we will develop models using stochastic processes which
will represent the events of division, death, acquisition of a
resistant mutation, and of neutral mutations (i.e., those that do not
affect individual growth) for each cell. Our interest lies in a
multi-scale context, wherein the initial sensitive population is large
and the probability of acquiring resistance is low.
Our aim is to study
the Site Frequency Spectrum (SFS), a tool for analysing the
distribution of neutral mutations within a population, which is
accessible through DNA sequencing. We are particularly interested in
neutral mutations shared by a significant proportion of the population,
a relatively understudied area. We aim to establish convergence in law
for the SFS, providing reliable predictions even with limited in vivo
or experimental data. Ultimately, we will develop statistical methods
to analyze such data. By achieving these objectives, we hope to enhance
our understanding of resistance mechanisms in cancer populations and
provide more efficient tools for predicting and analyzing treatment
outcomes.
|
|





