Our article is available to read at Inorg. Chem.
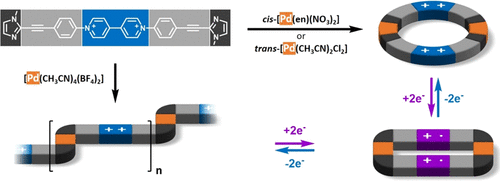
A metal-induced self-assembly strategy is used to promote the π-dimerization of viologen-based radicals at room temperature and in standard concentration ranges. Discrete box-shaped 2:2 (M:L) macrocycles or coordination polymers are formed in solution by self-assembly of a viologen-based ditopic ligand with cis-[Pd(en)(NO3)2], trans-[Pd(CH3CN)2(Cl)2], or [Pd(CH3CN)4(BF4)2]. Changing the redox state of the bipyridium units involved in the tectons, from their dicationic state to their radical cation state, results in a reversible “inflation/deflation” of the discrete 2:2 (M:L) macrocyclic assemblies associated to a large modification in the size of their inner cavity. Viologen-centered electron transfer is also used to trigger a dissociation of the coordination polymers formed with tetrakis(acetonitrile)Pd(II), the driving force of the disassembling process being the formation of discrete box-shaped 2:2 (M:L) assemblies stabilized by π-dimerization of both viologen cation radicals.
 Denis Frath obtained his PhD in chemistry from the University of Strasbourg in 2013 (laboratory of Dr. Gilles Ulrich and Raymond Ziessel). His doctoral work, supported by a MENRT scholarship, focused on hydrogen production using light and the synthesis of new fluorescent dyes. He then joined the Matsuda laboratory to work on photoresponsive self-assembled monolayers as a JSPS postdoctoral fellow at Kyoto University (2013-2015). After that, he worked on redox-active molecular layers for electronic devices at Paris Diderot University (2016-2017) and binuclear phthalocyanine metallic complexes for catalytic reactions at CNRS in Lyon (2018).
Denis Frath obtained his PhD in chemistry from the University of Strasbourg in 2013 (laboratory of Dr. Gilles Ulrich and Raymond Ziessel). His doctoral work, supported by a MENRT scholarship, focused on hydrogen production using light and the synthesis of new fluorescent dyes. He then joined the Matsuda laboratory to work on photoresponsive self-assembled monolayers as a JSPS postdoctoral fellow at Kyoto University (2013-2015). After that, he worked on redox-active molecular layers for electronic devices at Paris Diderot University (2016-2017) and binuclear phthalocyanine metallic complexes for catalytic reactions at CNRS in Lyon (2018).